Free Next Day Delivery Over £20 | Same Day Dispatch

What are TRPR and TPD regulations and how do they impact you? If you’re a vaper in the UK, understanding these rules is crucial.
They dictate tank capacity, E-Liquid bottle sizing, and nicotine strength for vaping products within the EU, and subsequently in the UK’s adapted TRPR post-Brexit. Dive into our comprehensive breakdown to stay informed.
The Tobacco Products Directive (TPD) was established with the aim of improving the functioning of the internal market for tobacco and related products, while ensuring high health protection for European citizens.

10ml Bottle Sizes

2ml Tank Capacity
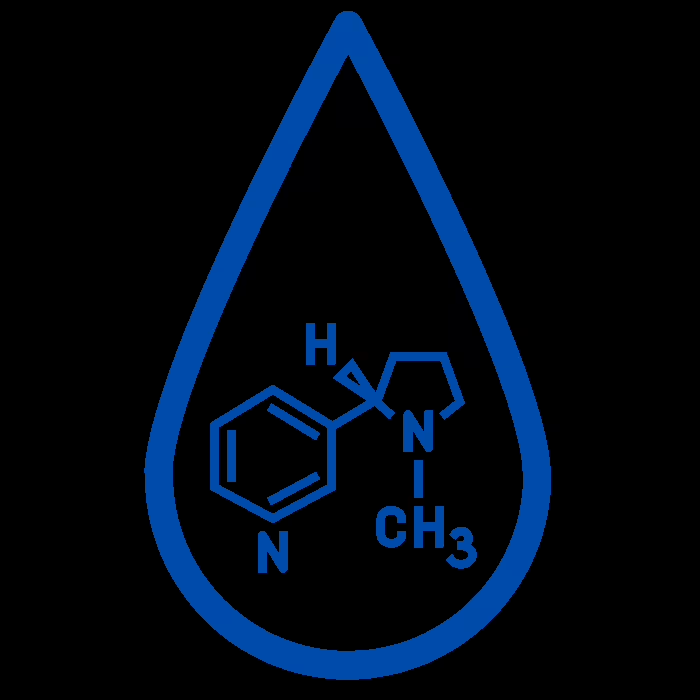
2% Max Nicotine

Product Certification
The TPD of 2014 introduced regulations for e-cigarettes, marking a significant stride in ensuring health protection and safety for consumers within the EU. These new regulations set safety and quality requirements for electronic cigarettes and required manufacturers to submit detailed product reports before placing them on the EU market.
The main points of TPD compliance revolve around restrictions on:
These regulations have significantly altered the consumer experience and imposed substantial compliance costs and red tape for UK manufacturers.

One of the pivotal stipulations of the TPD is the restriction on vape tank size. The directive restricts vape tank, pod, and cartridge capacity to no more than 2ml.
This regulation was implemented to ensure that electronic cigarettes and related products are used responsibly and that consumers are protected from excessive nicotine intake.
Despite the perceived limitations of this regulation, recalling its purpose is crucial. The reduction in tank size by the TPD aims to protect consumers, especially those new to vaping, from possible nicotine overexposure.
This restriction, however, has stimulated inventive adaptations among vaping enthusiasts to accommodate these new rules.
The updated TPD also imposed restrictions on the sizes of nicotine-containing E-Liquid bottles. Under these new regulations, E-Liquids containing nicotine must be sold in volumes of 10ml or less.
This means that, following the implementation of the TRPR regulations, individual E-Liquid bottles containing nicotine of 10ml will only be available in 10ml bottles.
These restrictions were designed to cover nicotine containing products and reduce the potential harm caused by accidental ingestion or skin exposure to nicotine, including nicotine containing E-Liquid.
However, they have also led to changes in how E-Liquids are packaged and sold, with manufacturers introducing ‘short fills’ that allow vapers to buy nicotine-free liquids in bulk and add their own nicotine.
Nicotine strength is another critical aspect regulated under the TPD. The directive stipulates that E-Liquids cannot contain more than 20mg/ml of nicotine. This regulation ensures a regulated upper limit for nicotine content in vaping products, protecting consumers from potential nicotine overexposure.
Although this rule may seem inhibiting, especially for heavy smokers transitioning to vaping, it is vital to bear in mind the motive behind it. The TPD’s establishment of a maximum nicotine strength is intended to curb nicotine addiction and encourage safer vaping habits.

Building on the foundation of the TPD, the UK developed the Tobacco & Related Products Regulation (TRPR), an equivalent legislation to adopt the TPD for nicotine-containing products.
The TRPR outlines the responsibilities of e-cigarette producers, covering the manufacturing, importing, or rebranding of vaping products for resale within the UK.
However, the TRPR deviates from the TPD in certain respects. For instance, it does not mandate an additional leaflet with health information if all necessary details are already displayed on the outer packaging. This deviation is indicative of the UK’s efforts to adapt the TPD regulations to better suit its specific market needs.
The Medicines and Healthcare products Regulatory Agency (MHRA) is instrumental in upholding the TRPR regulations in the UK. The MHRA is responsible for assessing and approving notifications of new tobacco products to ensure they comply with legal standards before being sold.
Post-Brexit, the MHRA has continued to regulate the market by actively updating lists of submitted and published e-cigarette and tobacco products, reflecting ongoing compliance efforts. This active oversight underscores the UK’s commitment to ensuring the safety and efficacy of vaping products.
Alongside these regulations, the TRPR mandates a leak-proof refill container mechanism for vaping products. This stipulation minimizes the risk of nicotine poisoning by preventing accidental exposure to E-Liquids during refilling.
Advancements in design and technology have resulted in vaping devices equipped with leak-proof refilling mechanisms. These devices not only comply with TRPR regulations but also offer a cleaner, safer, and more efficient vaping experience for consumers.
To ensure safety and transparency, the TRPR mandates emissions testing for all products. Manufacturers must conduct emissions testing on each flavour and flavour combination at least once.
This testing focuses on compounds such as acetaldehyde, acrolein, and formaldehyde, in accordance with how the product is expected to be used as per user instructions.
In addition, submitted toxicological data must detail all product components, specifically excluding prohibited carcinogens, CMR substances, or heavy metals from E-Liquids.
Notifications for e-cigarette products sent to the MHRA include exhaustive details like ingredients, emissions, and toxicological data, guaranteeing transparency and safety.

With the implementation of TPD and TRPR regulations, vapers have experienced significant changes. The capping of maximum nicotine strengths at 20mg/ml, limiting tanks to a maximum of 2ml, and restricting E-Liquid bottles to 10ml has notably altered the consumer experience.
Vape enthusiasts have devised ways to adjust to these changes. Some use expansion glass to ramp up tank capacity, while others have adopted short fill E-Liquids to circumvent the TPD’s 10ml restriction on nicotine-containing E-Liquids.
However, the restriction on E-Liquid nicotine strength to a maximum of 20mg/ml has sparked concern among heavy smokers about the potential difficulties in effectively transitioning to vaping.
The introduction of TPD and TRPR regulations has also posed challenges for E-Liquid manufacturers. These regulations have necessitated design changes, including child-resistant and tamper-evident packaging.
Detailed product notifications through the EU-CEG portal require extensive information on characteristics, formulation, toxicology, and emissions.
E-Liquid manufacturers have faced considerable compliance costs due to TPD regulations, including:
Furthermore, restrictions on advertising have also significantly impacted the E-Liquid industry.
What is ECID? ECID, or Electronic Cigarette Identification, is a unique identifier assigned to each e-liquid product placed on the market within the European Union. This identifier is mandated by the TPD and is vital for regulatory oversight and consumer safety.
Why is ECID Required? ECID serves several essential purposes, including ensuring consumer safety, transparency, and compliance with the TPD. It allows authorities to track and monitor vaping products effectively.
Dossier Preparation: Manufacturers must prepare a comprehensive dossier for each product, including detailed information about the product, ingredients, emissions data, manufacturing processes, and packaging.
Notification to Regulatory Authorities: Manufacturers are required to notify the relevant regulatory authorities in their EU member state regarding the intention to place a specific product on the market. This notification includes submitting the prepared dossier along with any necessary fees.
ECID Issuance: Upon successful review and approval of the dossier, regulatory authorities issue an ECID for the product. This identifier must be displayed prominently on the packaging and labelling.
Advertising bans for e-cigarettes were established under the EU Tobacco Products Directive Revision of 2016. These bans align e-cigarettes closer to the strictures for traditional tobacco products. The anti-promotion stipulations include:
The UK’s Tobacco and Related Products Regulations (TRPR) have further intensified advertising restrictions, complicating consumers’ search for information about vaping products. These restrictions limit promotional activities, making it more difficult for manufacturers to reach potential customers.
TPD and TRPR regulations have established regulatory controls for nicotine-containing e-cigarettes and refill containers that emphasize product safety, quality, and supporting informed consumer choices in smoke free environments.
The MHRA actively oversees the risks linked to tobacco products by handling adverse event reports and flaws in products to safeguard public health.
Safety measures, such as testing and reporting E-Liquid ingredients, have helped protect vapers from conditions like EVALI.
Additionally, TRPR requires the disclosure of metal emissions from vaping hardware. These measures underscore the commitment of regulatory bodies to protect public health and ensure the safety and quality of vaping products.
Following the conclusion of the Brexit transition period, the UK government has delineated forthcoming modifications to e-cigarette regulations. The UK has preserved many TPD regulations post-Brexit but now has the flexibility to enact amendments that could be more tailored to the needs of the UK.
A series of updates have been released documenting specific changes in the UK’s e-cigarette regulations after Brexit, indicating active regulation management.
Future changes to UK vaping regulations will likely focus on meeting the UK’s market specifics and responding to technological and public health advancements.
Loading FAQAccordion...
In conclusion, the TPD and the UK’s adaptation of it through the TRPR have had significant implications for vapers and E-Liquid manufacturers alike. From vape tank size restrictions to E-Liquid bottle size limitations, from maximum nicotine strength to advertising restrictions, these regulations have reshaped the vaping landscape in the UK.
While these regulations may pose challenges in the short term, they aim to safeguard public health and ensure the safety and quality of vaping products.
As the UK continues to adapt these regulations post-Brexit, it will be crucial to strike a balance between ensuring safety and accommodating the needs of vapers and manufacturers.

Neil is a sub-ohm vaping aficionado and wordsmith extraordinaire, aged 25. With his insatiable passion for vaping and his massive collection of vape mods, Neil weaves his expertise into every article he crafts. When he’s not writing, he's exploring new flavours, trying out vape tricks, or riding his bike. Get ready for an exciting journey through the clouds with Neil as your guide!
Yes, the UK TPD is regulated through the UK Tobacco and Related Products Regulations 2016.